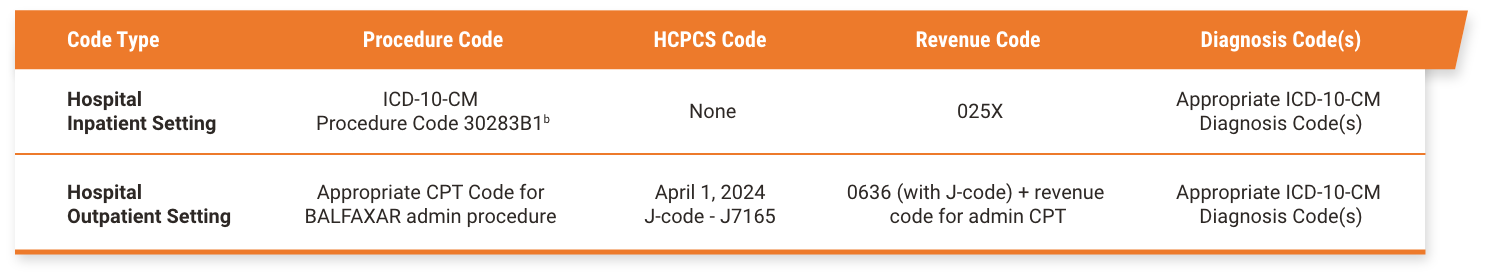
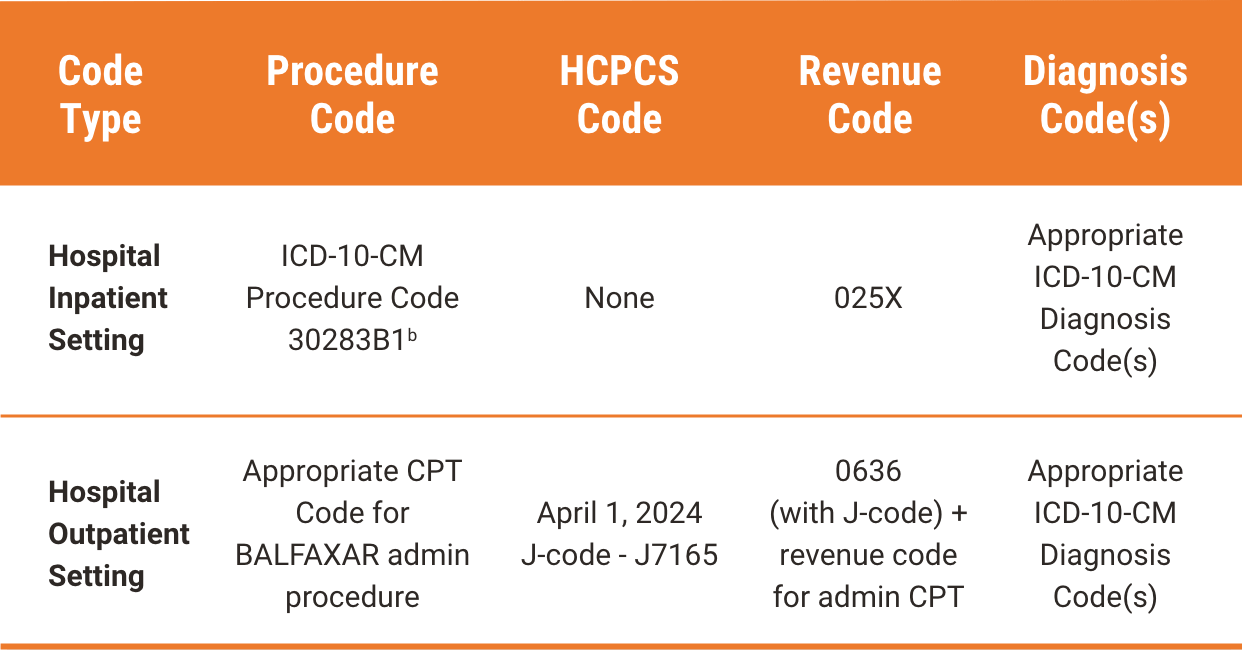
BALFAXAR Reimbursement
Learn More About Total Support With BALFAXAR
aThe treating physician is solely responsible for the determination of the appropriate ICD-10-CM diagnosis codes that describe the patient’s condition and are supported by the medical record. All codes provided are for informational purposes and do not constitute an exhaustive list. The provided CPT®, HCPCS, ICD-10-CM, and ICD-10-PCS codes are based on AMA or CMS guidelines. The billing party is solely responsible for the coding of services. Please verify current coding requirements directly with the payer being billed as government and other third-party payer coding requirements may change.
bInfusion of 4F-PCC.
Reference: 1. CMS.gov. Inpatient Prospective Payment System (IPPS). Updated October 19, 2023. Accessed October 31, 2023. https://www.cms.gov/cms-guide-medical-technology-companies-and-other-interested-parties/payment/ipps
Important Safety InformationView More +
WARNING: ARTERIAL AND VENOUS THROMBOEMBOLIC COMPLICATIONS
Patients being treated with Vitamin K antagonists (VKA) therapy have underlying disease states that predispose them to thromboembolic events. Potential benefits of reversing VKA should be weighed against the potential risks of thromboembolic events, especially in patients with the history of a thromboembolic
Important Safety InformationView Less —
WARNING: ARTERIAL AND VENOUS THROMBOEMBOLIC COMPLICATIONS
Patients being treated with Vitamin K antagonists (VKA) therapy have underlying disease states that predispose them to thromboembolic events. Potential benefits of reversing VKA should be weighed against the potential risks of thromboembolic events, especially in patients with the history of a thromboembolic event. Resumption of anticoagulation should be carefully considered as soon as the risk of thromboembolic events outweighs the risk of acute bleeding. Both fatal and non-fatal arterial and venous thromboembolic complications have been reported with BALFAXAR in clinical trials and post marketing surveillance. Monitor patients receiving BALFAXAR for signs and symptoms of thromboembolic events. BALFAXAR may not be suitable in patients with thromboembolic events in the prior 3 months.
BALFAXAR is contraindicated in patients with known anaphylactic or severe systemic reactions to BALFAXAR or any of its components. BALFAXAR is also contraindicated in patients with a known allergy to heparin, a history of heparin-induced thrombocytopenia (HIT), and IgA deficient patients with known antibodies against IgA.
In clinical trials, the most frequent (≥3%) adverse reactions observed in subjects receiving BALFAXAR were procedural pain, wound complications, asthenia, anemia, dysuria, procedural vomiting, and catheter-site-related reaction.
BALFAXAR is derived from human plasma. The risk of transmission of infectious agents, including viruses and, theoretically, the Creutzfeldt-Jakob disease (CJD) agent and its variant (vCJD), cannot be completely eliminated.
Indications
BALFAXAR (prothrombin complex concentrate, human-lans) is a blood coagulation factor replacement product indicated for the urgent reversal of acquired coagulation factor deficiency induced by Vitamin K antagonist (VKA, e.g., warfarin) therapy in adult patients with need for an urgent surgery/invasive procedure.
Please click here for Full Prescribing Information, including BOXED WARNING.
To report suspected adverse reactions, contact Octapharma USA, Inc. at 1-866-766-4860 or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.